Cost Analysis of Hemiarthroplasty Versus Reverse Shoulder Arthroplasty for Fractures
Jason A. Solomon, MD; Sheeba M. Joseph, MD, MS; Yousef Shishani, MD; Brian N. Victoroff, MD; John H. Wilber, MD; Reuben Gobezie, MD; Robert J. Gillespie, MD
Abstract
View / Download, PDF Version of this Article
Complex proximal humerus fractures in older patients can be treated with hemiarthroplasty (HA) or reverse shoulder arthroplasty (RSA), with both providing good pain relief and function. This study compared the costs, complications, and outcomes of HA vs RSA after proximal humerus fracture in older patients. Patients 65 years or older who were admitted between January 2007 and August 2011 with a 3 or 4 part proximal humerus fracture and treated with HA or RSA were identified. Surgeries were performed at the same institution by 1 of 3 surgeons trained in trauma or shoulder surgery. Operating room costs, implant costs, total costs to the patient and hospital, and range of motion were compared. In the study group, 8 patients (7 women and 1 man; mean age, 77 years) received HA and 16 patients (13 women and 3 men; mean age, 77 years) received RSA. Hemiarthroplasty implant cost and operating room cost were $9140 and $8900 less than those of RSA, respectively (P<.001). The total cost to the patient was $33,480 for HA vs $57,000 for RSA (P<.001) with no difference in admission length, transfusion requirements, or final range of motion. In patients with complex proximal humerus fractures, RSA restored function similar to HA and resulted in better pain and outcome scores. However, RSA had a significantly higher cost to both the patient and the hospital compared with HA. Further investigation of postsurgical rehabilitation costs, skilled nursing needs, or revision surgery will elucidate whether there is long-term functional or financial benefits to RSA over HA. [Orthopedics. 2016; 39(4):230-234.]
The treatment of complex proximal humerus fractures in older patients remains controversial. As the third most common osteoporotic fracture, proximal humerus fractures represent a significant cost to the Medicare system.1 Seventy percent of all proximal humeral fractures occur in patients older than 60 years2. The Neer classification of proximal humerus fractures depends on the number of fragments, with a fragment de ned as being greater than 1 cm displaced or greater than 45° angulated.3 Three and 4-part proximal humerus fractures may be treated nonoperatively, with closed reduction and percutaneous pinning, with open reduction and internal fixation, or with prosthetic replacement.3 Although the majority of fractures may be treated nonoperatively, severe fracture patterns can often lead to poor functional outcomes.
Complex proximal humerus fractures that may require arthroplasty include fracture-dislocations, comminuted head splitting fractures, and complex fracture patterns with significant risk of avascular necrosis.4 Treatment of such fractures in older patients with hemiarthroplasty (HA) has had inconsistent results due to failure of tuberosity healing, continued pain, loss of motion, infection, and posttraumatic glenoid degeneration.5-8 Recently, treatment with reverse shoulder arthroplasty (RSA) has been shown to provide good pain relief and restoration of function with complication rates that compare favorably with alternative treatments.9-11
The issue of rising costs related to surgical outcomes continues to be a main focus in health care. Justification for the increased cost of RSA has come into question recently despite its proposed benefit to improving pain and function compared with HA.11 This study compared (1) range of motion and American Shoulder and Elbow Surgeons (ASES) total and pain scores, (2) rate and cost of complications, (3) charges to the patient, and (4) charges and reimbursement to the hospital system in patients with 3and 4part proximal humerus fractures treated with arthroplasty. The current authors hypothesized that both the hospital and the patient would experience higher costs of care associated with RSA, but patients receiving this treatment would have improved range of motion, pain, and outcome scores compared with patients treated with HA.
Materials and Methods
After receiving institutional review board approval, a retrospective chart review was performed. The institution’s Medicare database was queried for shoulder arthroplasty procedures performed for proximal humerus fractures in patients 65 years or older from January 2007 to August 2011. Patients with 3 or 4 part proximal humeral fractures treated with either HA or RSA were included in the study. All arthroplasties were performed at a single institution by 1 of 3 surgeons (1 surgeon trained in trauma surgery and 2 surgeons trained in shoulder surgery).
During the study period, 24 patients underwent shoulder arthroplasty for a proximal humerus fracture (20 women and 4 men). After reviewing the patients’ medical comorbidities, level of function, and preoperative radiographs, a determination was made by the treating physician for HA or RSA. The surgeons’ training and comfort with implant options also contributed to their decision-making process as the surgeon trained in trauma surgery did not perform any RSAs. The ultimate indication for using RSA rather than HA was based on the patient’s prior history of rotator cuff disease, severe tuberosity comminution preventing reconstruction, or radiographic evidence of rotator cuff arthropathy.
Outcomes were assessed at final follow-up with ASES total and pain scores, and range-of-motion (ROM) measurements. The All Patient Re ned (APR) mortality index was calculated for each patient to compare operative risk and severity of illness. Length of hospital stay and packed red blood cell transfusion requirements also were recorded. Imaging was obtained preoperatively and postoperatively, and patients were evaluated at 1 week, 1 month, 3 months, and at final follow-up (18 to 73 months) after the initial operation.
All surgeries were performed in the beach chair position with prosthesis placement via the standard deltopectoral approach. The greater and lesser tuberosity fracture fragments were identified and tagged for later reduction and repair. The biceps tendon was routinely tenodesed to the pectoralis major tendon. Two drill holes were made in the humeral shaft for subsequently securing the tagged tuberosity fragments.
The Bigliani/Flatow hemiarthroplasty prosthesis (Zimmer, Warsaw, Indiana) and the Aequalis reverse shoulder system (Tornier, Bloomington, Minnesota) were used for HA and RSA, respectively. All implants were cemented and placed in 20° of retroversion relative to the trans-epicondylar axis. Cerclage sutures were placed around the inferomedial border of the prosthesis as described by Boileau et al.12
Postoperatively, patients were in a sling for 3 weeks while allowing active elbow, wrist, and hand range of motion. Patients began pendulum exercises at 1 week and shoulder active range of motion at 3 to 4 weeks. All patients participated in physical therapy postoperatively.
Cost data were retrieved from the hospital’s Medicare database. Implant manufacturers were contacted to con rm implant prices during the study period. All physical therapy was performed within the study site’s hospital system.
Statistical Analysis
Data were analyzed with parametric and nonparametric tests using SPSS (IBM Corp, Armonk, New York). The aforementioned outcomes measured and the surgery-related parameters and costs were compared between HA and RSA with significance de ned as P<.05.
Results
In this retrospective study, 8 patients underwent HA and 16 patients underwent RSA. Both groups of patients were similar in average age, sex, and complication rate (Table). All patients survived to final follow-up. Mean length of clinical follow-up was 43 months (range, 18-73 months). Mean age at the time of injury was 77 years (range, 68-96 years) in the HA group and 77 years (range, 65-88 years) in the RSA group (P=.94). All fractures were the result of a low-energy fall.
As demonstrated in Figure 1, mean forward flexion was 104° (95% confidence interval [CI], 95°-110°) in the HA group and 110° (95% CI, 100°-130°) in the RSA group (P=.34). There was no difference in mean external rotation between the HA group (29°, 95% CI, 22°-35°) and the RSA group (28°, 95% CI, 25°-30°) (P=.80).
Patients in the HA group had significantly lower ASES scores as well as pain scores compared with the RSA group (Figure 2). Mean ASES score was 59 (95% CI, 47-71) in the HA group compared with 79 (95% CI, 72-86) in the RSA group (P=.020). Mean ASES pain score was 32 (95% CI, 22-34) in the HA group compared with 42 (95% CI, 39-45) in the RSA group (P=.025).
There was no difference in length of hospital stay between the HA group (4.4 days, 95% CI, 3.5-5.3) and the RSA group (3.6 days, 95% CI, 2.8-4.3) (P=.26) (Figure 3). There was no difference in need for blood transfusion in the HA group (0.88 units, 95% CI, 0.47-1.3) compared with the RSA group (0.5 units, 95% CI, .14-0.86) (P=.27). There also was no difference in the 1-year mortality rate between the HA and RSA groups. Seven of 8 patients in the HA group required transfer to a skilled nursing facility following inpatient admission compared with 9 of 16 patients in the RSA group.
Study Group Characteristics and Complication Rates
| Characteristic | Hemiarthroplasty | Reverse Shoulder Arthroplasty | P |
| Age, mean (range), y | 77 (68-96) | 77 (65-88) | .94 |
| Women | 89% | 81% | .70 |
| Mechanism of injury | Low-energy fall | Low-energy fall | – |
| Complications, No. | 1 | 2 | 1 |
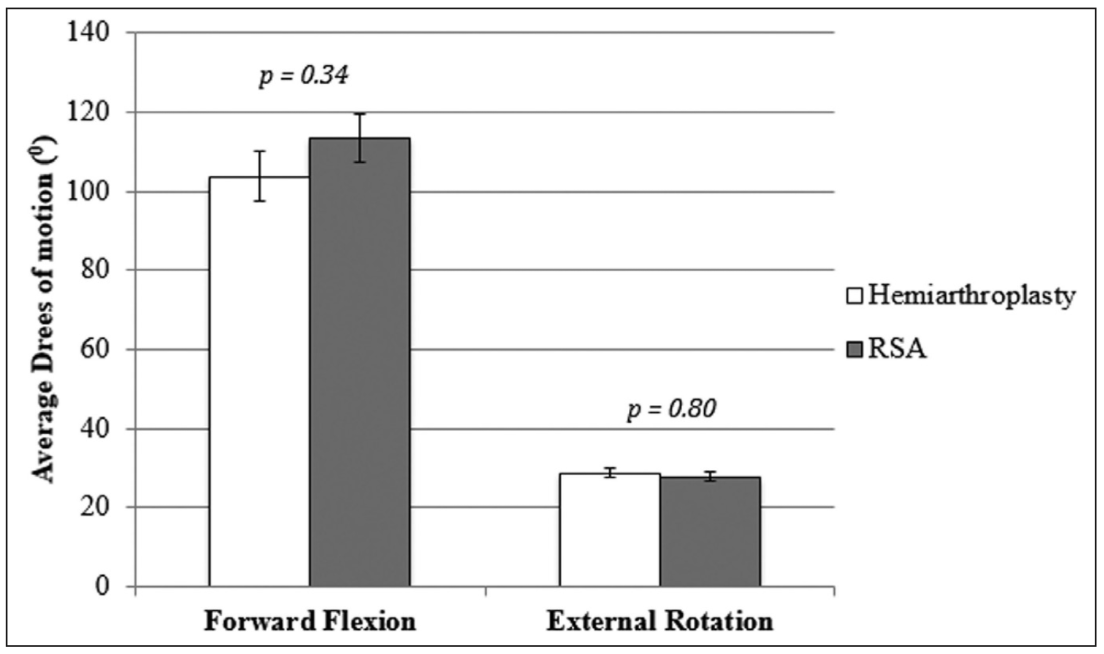
Figure 1: Graph comparing mean range of motion (degrees) for forward exion and external rotation between patients receiving hemiarthroplasty vs reverse shoulder arthroplasty (RSA) for complex proximal humerus fractures.
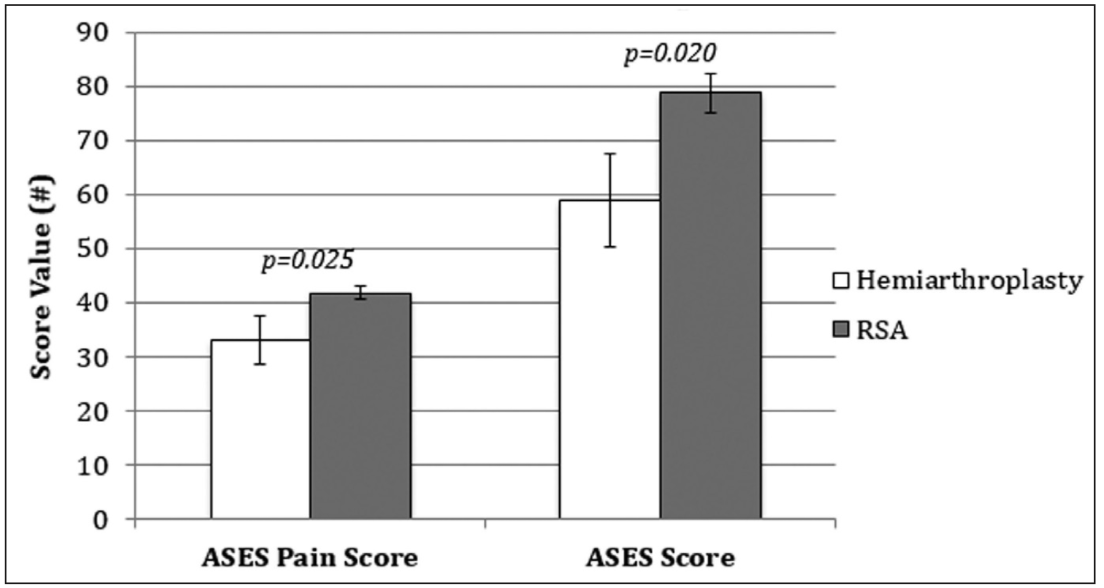
Figure 2: Graph comparing mean American Shoulder and Elbow Surgeons (ASES) pain scores and ASES scores for patients undergoing hemiarthroplasty and reverse shoulder arthroplasty (RSA) for complex proximal humerus fractures.
Radiographic Analysis
There were 2 incidences of greater tuberosity nonunion in the HA group; 1 of these patients progressed to anterosuperior escape. Neither of these patients has elected to undergo revision arthroplasty. In the RSA group, there was 1 incidence of scapular notching that was classified as grade 2 according to the Nerot-Sirveaux classification at final follow-up. There was no humeral implant loosening in either group.
Cost Analysis
During the study time period, the mean cost of HA and RSA prostheses was $4160 and $13,300, respectively (Figure 4). The total cost to the hospital included operating room costs, implant costs, inpatient physical therapy, and admission costs. This was significantly lower in the HA group than in the RSA group ($22,000 and $40,000, respectively; P=.001). The total charge to the patient also was significantly lower in the HA group compared with the RSA group ($33,480 and $57,000, respectively; P=.001). There was no difference in mean reimbursement to the hospital system for the admission between the HA and RSA groups ($11,200 and $13,200, respectively; P=.14). There also was no difference in inpatient physical therapy costs between the HA and RSA groups ($224 and $268, respectively; P=.48).
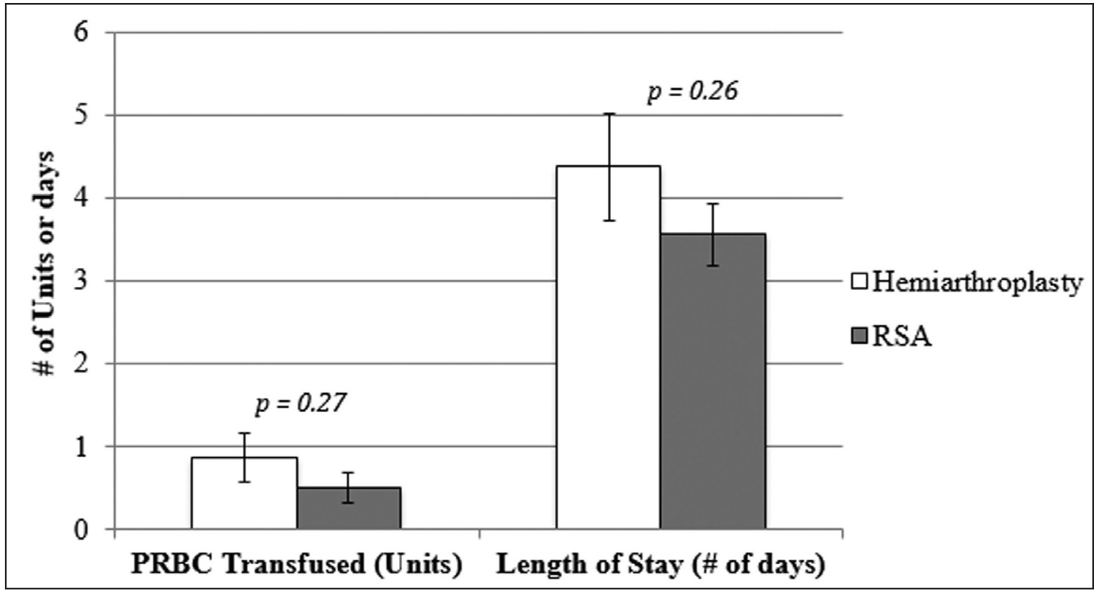
Figure 2: Graph comparing length of stay (days) and units of blood transfused postoperatively for patients undergoing hemiarthroplasty and reverse shoulder arthroplasty (RSA) for complex proximal humerus fractures. Abbreviation: PRBC, packed red blood cells.
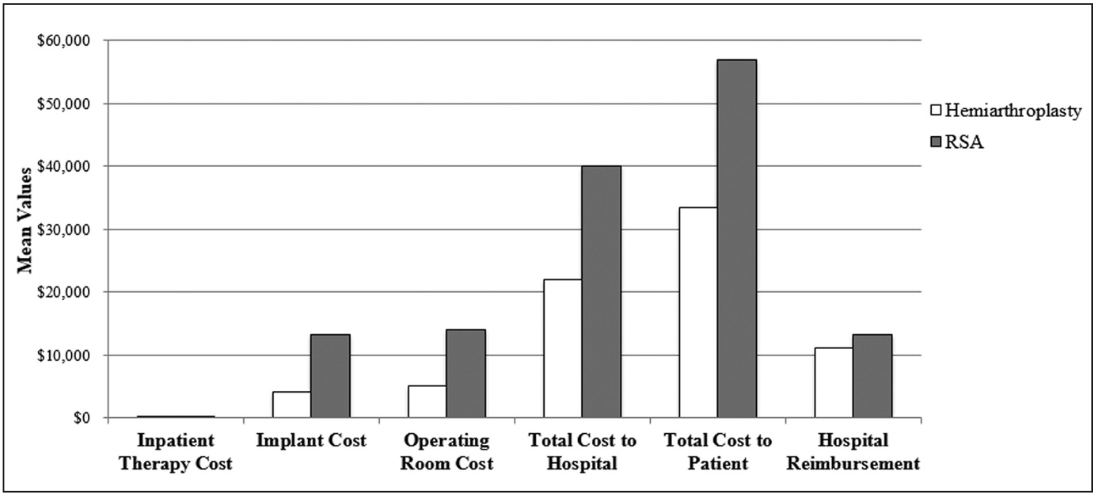
Figure 2: Graph comparing costs for various expenses incurred in patients undergoing hemiarthroplasty vs reverse shoulder arthroplasty (RSA) for complex proximal humerus fractures.
Complications
The All Patient Re ned Diagnosis Related Groups (APR-DRG) was used to stratify level of illness and risk of mortality on a scale of 1 to 4. The APR risk was 1.6 (95% CI, 4.5-7.6) in the HA group and 1.3 (95% CI, 1.0-1.6) in the RSA group, indicating no significant difference in medical risk factors for perioperative related complications between the 2 groups (P=.20).
Additional surgical procedures were required in 3 patients. In the HA group, a 65-year-old woman developed a postoperative infection within 2 weeks of surgery; she required readmission for open irrigation and debridement. This patient was a poorly controlled diabetic with a hemoglobin A1C of 9.1%. The total cost to the hospital for this readmission was $19,995.
In the RSA group, 2 patients sustained complications. A 67-year-old man fell out of bed on the night of surgery and sustained a dislocation. This required an operative open reduction with exchange to a larger polyethylene liner. The second patient, a 79-year-old woman, presented to the clinic 2 weeks following her surgery with a dislocated shoulder that had occurred during physical therapy. She required an open reduction and exchange to a larger polyethylene liner. The total costs of readmission for these complications were $13,255 and $13,600, respectively.
Discussion
There continues to be controversy in the selection of treatment for complex proximal humerus fractures requiring arthroplasty. Given the significant cost differential between these 2 treatments, this study compared HA with RSA for complex proximal humerus fractures in terms of postoperative outcomes and cost to the patient and hospital. This study found that despite an increased cost to the hospital and to the patient, RSA improved pain relief and outcome scores compared with HA in the treatment of older patients (≥65 years) with 3 or 4 part proximal humerus fractures.
Using shoulder arthroplasty to treat older patients with complex proximal humeral fractures and osteopenic bone is difficult. Antuña et al5 retrospectively reviewed 57 patients (mean age, 66 years) who underwent HA to treat acute proximal humeral fractures with a minimum follow-up of 5 years (mean, 10.3 years). They found that 30 of 57 patients had unsatisfactory outcomes according to a modified Neer result rating system, and 16% had moderate to severe pain at final follow-up.5 Most of the poor results were attributed to poor rotator cuff function or nonunion of the tuberosities.5
Boyle et al9 performed a retrospective review using the New Zealand Joint Registry to compare 55 patients who underwent HA with 313 patients who underwent RSA for fracture management at 6 months and 5 years following surgery. The RSA group had significantly improved Oxford shoulder scores at 5 years compared with HA, but no difference in revision rate or 1-year mortality rate.9 In the current study of older patients with equivalent APR mortality risk, RSA restored range of motion to the same level as HA but patients who underwent RSA also had improved pain and outcome scores.
The use of RSA has the theoretical benefit of relying less on rotator cuff integrity and tuberosity union, and it has shown good results when used for glenohumeral arthritis, rotator cuff deficiency, and pseudoparalysis. In a systematic review of 15 studies with 377 patients treated with RSA and 504 patients treated with HA for proximal humeral fractures in older patients, MataFink et al13 found patients who underwent RSA had improved forward flexion and outcomes scores, but diminished external rotation, with similar complication rates. In a retrospective review of 32 patients older than 65 years who underwent RSA for acute proximal humeral fractures, Lenarz et al11 found patients attained a mean active forward elevation of 139° and a mean visual analog pain score of 1.1, with no evidence of component loosening or need for reoperation. In a meta-analysis of 9 studies that included a total of 247 patients, Anakwenze et al14 found RSA treatment of proximal humerus fractures had improved external rotation with tuberosity repair leading to an average forward flexion of 113° and average external rotation of 26°.
The impact of an older patient sustaining a proximal humeral fracture can be significant, both to the patient as well as to the health system. Jain et al15 showed lower costs associated with treatment of proximal humerus fractures have been found to correlate with surgeon volume (≥20 shoulder arthroplasties performed per year), open reduction and internal fixation over HA, and fewer comorbidities.15 Ponce et al16 reviewed the 2011 Nationwide Inpatient Sample database and found RSA to be an independent risk factor for increased inpatient morbidity, mortality, and hospital costs compared with anatomic total shoulder arthroplasty.
In the current study, costs to both the hospital and the patient were significantly greater in the RSA group compared with the HA group. At a mean of 43 months of follow-up, no patient in the HA group had required revision arthroplasty to a reverse prosthesis due to pain or loss of motion. Complications occurring within 30 days of surgery incurred a significant additional financial burden to the hospital that approached $20,000 in the HA group and averaged $13,428 in the RSA group. In the HA group, the hospital was reimbursed 51% of its total cost and 33% of its total charges. In the RSA group, the hospital was reimbursed 33% of its total cost and 23% of its total charges. Overall, during this period, the hospital was unable to recover the cost of treatment for HA and RSA.
Limitations
This study has several limitations. It is a retrospective study at a single institution with surgery performed by orthopedic surgeons from 2 subspecialties including traumatology and shoulder surgery. As such, there may have been training dependent bias toward treatment with HA or RSA. In addition, the sample size was relatively small. Longer follow-up may reveal further complications including need for conversion of HA to RSA10,17 or failure of the reverse prosthesis from loosening, scapular notching, or fracture.
Conclusion
Three and 4-part proximal humerus fractures in older patients may be managed with HA or RSA. Although this study found the cost of RSA to the hospital and the patient exceeded that of HA, the pain relief and outcome scores were significantly improved with RSA, and the complication rates of RSA were similar with those of HA. Future work examining longer-term follow-up may give a different representation of complications and related costs for HA vs RSA. Finally, the increased use of RSA and subsequent decreased cost that is occurring in the US market may make its use less controversial when appropriately indicated for the treatment of complex proximal humerus fractures.
References
- Baron JA, Karagas M, Barrett J, et al. Basic epidemiology of fractures of the upper and lower limb among Americans over 65 years of age. Epidemiology. 1996; 7(6):612-618.
- Lind T, Krøner K, Jensen J. The epidemiology of fractures of the proximal humerus. Arch Orthop Trauma Surg. 1989; 108(5):285-287.
- Neer CS II. Displaced proximal humeral fractures: I. Classi cation and evaluation. J Bone Joint Surg Am. 1970; 52(6):1077-1089.
- Boileau P, Walch G, Trojani C, Sinnerton R, Romeo AA, Veneau B. Sequelae of fractures of the proximal humerus: surgical classification and limits of shoulder arthroplasty. In: Walch G, Boileau P, eds. Shoulder Arthroplasty. Berlin, Germany: Springer-Verlag; 1999:349-358.
- Antuña SA, Sperling JW, Cofield RH. Shoulder hemiarthroplasty for acute fractures of the proximal humerus: a minimum five-year follow-up. J Shoulder Elbow Surg. 2008; 17(2):202-209.
- Boileau P, Krishnan SG, Tinsi L, Walch G, Coste JS, Molé D. Tuberosity malposition and migration: reasons for poor outcomes after hemiarthroplasty for displaced fractures of the proximal humerus. J Shoulder Elbow Surg. 2002; 11(5):401-412.
- Grönhagen CM, Abbaszadegan H, Révay SA, Adolphson PY. Medium-term results after primary hemiarthroplasty for comminute proximal humerus fractures: a study of 46 patients followed up for an average of 4.4 years. J Shoulder Elbow Surg. 2007; 16(6):766-773.
- Noyes MP, Kleinhenz B, Markert RJ, Crosby LA. Functional and radiographic long-term outcomes of hemiarthroplasty for proximal humeral fractures. J Shoulder Elbow Surg. 2011; 20(3):372-377.
- Boyle MJ, Youn SM, Frampton CM, Ball CM. Functional outcomes of reverse shoulder arthroplasty compared with hemiarthroplasty for acute proximal humeral fractures. J Shoulder Elbow Surg. 2013; 22(1):32-37.
- Cuff DJ, Pupello DR. Comparison of hemiarthroplasty and reverse shoulder arthroplasty for the treatment of proximal humeral fractures in elderly patients. J Bone Joint Surg Am. 2013; 95(22):2050-2055.
- Lenarz C, Shishani Y, McCrum C, Nowinski RJ, Edwards TB, Gobezie R. Is reverse shoulder arthroplasty appropriate for the treatment of fractures in the older patient? Early observations. Clin Orthop Relat Res. 2011; 469(12):3324-3331.
- Boileau P, Trojani C, Walch G, Krishnan SG, Romeo A, Sinnerton R. Shoulder arthroplasty for the treatment of the sequelae of fractures of the proximal humerus. J Shoulder Elbow Surg. 2001; 10(4):299-308.
- Mata-Fink A, Meinke M, Jones C, Kim B, Bell JE. Reverse shoulder arthroplasty for treatment of proximal humeral fractures in older adults: a systematic review. J Shoulder Elbow Surg. 2013; 22(12):1737-1748.
- Anakwenze OA, Zoller S, Ahmad CS, Levine WN. Reverse shoulder arthroplasty for acute proximal humerus fractures: a systematic review. J Shoulder Elbow Surg. 2014; 23(4):e73e80.
- Jain NB, Kuye I, Higgins LD, Warner JJ. Surgeon volume is associated with cost and variation in surgical treatment of proximal humeral fractures. Clin Orthop Relat Res. 2013; 471(2):655-664.
- Ponce BA, Oladeji LO, Rogers ME, Menendez ME. Comparative analysis of anatomic and reverse total shoulder arthroplasty: in-hospital outcomes and costs. J Shoulder Elbow Surg. 2015; 24(3):460-467.
- Levy JC, Virani N, Pupello D, Frankle M. Use of the reverse shoulder prosthesis for the treatment of failed hemiarthroplasty in patients with glenohumeral arthritis and rotator cuff deficiency. J Bone Joint Surg Br. 2007; 89(2):189-195.
The authors are from the Department of Orthopaedic Surgery (JAS, SMJ, BNV, RJG), University Hospitals Case Medical Center, and the Department of Orthopaedic Surgery (JHW), MetroHealth Medical Center, Cleveland; and the Cleveland Shoulder Institute (YS, RG), May eld Heights, Ohio.
Drs Solomon, Joseph, Shishani, Victoroff, and Wilber have no relevant financial relationships to disclose. Dr Gobezie is a paid consultant for Arthrex Inc. Dr Gillespie is a paid consultant for Tornier and receives teaching fees from DJO and Tornier.
Correspondence should be addressed to: Robert J. Gillespie, MD, Department of Orthopaedic Surgery, University Hospitals Case Medical Center, Hanna House 6, 11000 Euclid Ave, Cleveland, OH 44106.
Received: August 31, 2015; Accepted: November 25, 2015.
doi: 10.3928/01477447-20160610-03


